Harold Cole Watkins, PhD, overcome with remorse, killed himself one rainy night in late 1937. A few months earlier, Dr. Watkins was on cloud nine. His new, sweet, raspberry-flavored, Elixir Sulfanilamide made it possible for parents to administer the bitter sulfa medicine to their children sick with Streptococcus infections, commonly known as strep throat. A few months earlier, Dr. Watkins was on cloud nine. His new, sweet, raspberry-flavored, Elixir Sulfanilamide made it possible for parents to administer the bitter sulfa medicine to their children sick with Streptococcus infections. Sore throats.
A pharmacist employed by S.E. Maassengil Co., Dr. Watkins met the company’s goal in response to public demand for a liquid form of the hard to swallow pill. Now, over a hundred people were dead, most of them children. Some children died in their mother’s
arms, within minutes of taking the medicine that was supposed to cure them. Some lingered for days in excruciating pain, as their kidneys failed.
Dr. Watkins was no monster. He was just a chemist looking for a formula that would dissolve the newest miracle drug. Sulfa won’t dissolve in water, but adding a small amount of the sweet tasting diethylene glycol makes the drug goes into solution. Thousands of bottles of poison. 633 shipments, went out across the country after passing tests for flavor, odor, and appearance.
Dr. Watkins, convinced of his innocence, took the medication himself, fell ill, and then recuperated. Massengil tried to recover the shipments, but records were scanty and incomplete at best. In some cases, pharmacists sold the Elixir without a prescription. One pharmacist had 49 prescriptions with identification like “Betty, 9 months old.” In other cases, physicians afraid of recrimination, refused to reveal their patients’ names.
Out of the graves of these children rose the strength of FDA. In 1938, Congress at last revised the Food, Drug &Cosmetic (FD&C) Act. The original FD&C Act required purity of our Foods, Drugs, and Cosmetics, but had little teeth to enforce even that requirement. The 1938 revision demanded testing to prove safety.
The review and approval process FDA requires is successful at preventing unsafe drugs from reaching the market. Thalidomide, a favorite “mother’s helper” sleep agent, resulted in thousands of babies born with disfigured limbs in Germany and Europe.
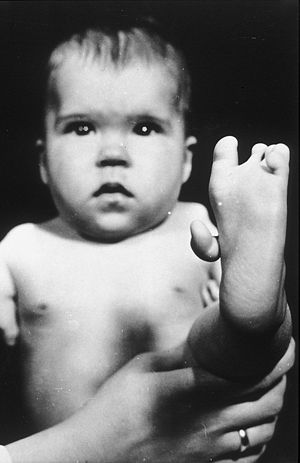
Very few babies with “flipper-limbs” were born in the US because FDA refused to approve Thalidomide. Instead, approval got held up in red tape and repeated studies, as FDA requested additional safety data. Some people, impatient with FDA snail-like pace, opted to get Thalidomide on the black market. Some of us, who grew up in the sixties, had a friend with a shriveled arm, perhaps a little finger protruding from the useless appendage, or maybe a classmate with no legs at all. Following this tragedy, the FD&C act was amended once again. Now drugs must be proven effective, as well as safe and pure.
Today, American people trust that the medication they take is safe, pure, and effective, and will prevent, treat, or cure what troubles them. But this security has exacted its pound of flesh.
Safety studies grow more and more complex. As scientists learn more about the variability of the human condition, the intricacies of safety studies grow. It is not enough to know the effect in a large number of people. Race, gender, and age must be studied. Possible drug interactions, and even variation in personal diet must be considered.
Due in part to persistent efforts of ethical pharmaceutical companies and the cautious nature of FDA, life expectancy in this country increased by 70% since the Elixir Sulfanilamide tragedy. Our quality of life improved, due to effective medications for chronic diseases like diabetes, high blood pressure, arthritis, and even Alzheimer’s disease.

graph showing differences in release kinetics between immediate drug
release, sustained drug release and controlled drug release
pharmaceutical formulations (Photo credit: Wikipedia)
It now takes 10-15 years, and more than $800 million dollars in research and development before a pharmaceutical product gets to market. For every drug that’s approved, 5,000 are not. Only three of every ten drugs earn enough to cover the development cost. No wonder our medicine costs so much.
But the cost of safe and effective medicine doesn’t stop with approval. Once a drug gets approved, controls must be put in place to assure the pharmaceutical product is manufactured consistently, safely labeled and packaged, and stored in a way that maintains the integrity of the medicine. Suppliers are inspected and quality agreements put in place. Ingredients are tested to exacting standards. Manufacturing steps are checked and cross-checked to make sure no mistakes go undetected. Even equipment cleaning is verified, so there is no chance that traces of another medicine or a cleaning chemical might put a patient in danger.
Unfortunately, no amount of testing, checking, and documenting can completely thwart the ne’r-do-well, the greedy, or the ignorant. Well-meaning people, trying to save money, order unapproved medicines through untraceable web-addresses. In an effort to cut costs, Manufacturers go to third world nations, many with ill-defined controls in place. We are in danger of repeating history. Incidences like toothpaste formulated with diethylene glycol (the same ingredient found in anti-freeze), or milk contaminated with melamine (to enhance the protein level) are eerie in their familiarity. Today most manufacturing sites for nutritional supplements lack manufacturing and testing controls. At the same time, I hear discussions that soon new drugs may be required to show not only safety, purity, and effectiveness, but superiority to existing drugs. I can only imagine what that testing might look like.
Whether the fault is greed, ignorance, or lack of respect for human life, will we not learn from our past mistakes? Everyone wants less regulation. Unless the regulation serves their needs. Right? How much is too much? What is the solution?



